- Center on Health Equity & Access
- Clinical
- Health Care Cost
- Health Care Delivery
- Insurance
- Policy
- Technology
- Value-Based Care
The Evolving Treatment Landscape of Pulmonary Arterial Hypertension
Abstract
Pulmonary arterial hypertension (PAH) is a severe disease with poor prognosis and shortened life expectancy. Treatment has traditionally involved the sequential use of endothelin receptor agonists, prostacyclin therapies, and nitric oxide pathway modulators, which each have distinct mechanisms of action leading to pulmonary vasodilation, and improvement in exercise capacity, hemodynamic measures, and clinical outcomes for patients with PAH. This article provides a review of goals of therapy in PAH, determinants of prognosis and levels of patient risk, and additional factors that guide treatment decision making. Recent research in combination therapies has created a paradigm shift in the treatment of PAH and will be reviewed. Additionally, recent updates to the American College of Chest Physicians guidelines will be reviewed along with the updated evidence-based treatment algorithm. Finally, trial data will be evaluated for the recently developed agent selexipag and improved treprostinil delivery formulations that may provide enhanced convenience.
Am J Manag Care. 2021;27(3):S42-S52. https://doi.org/10.37765/ajmc.2021.88610
Goals of Therapy in Pulmonary Arterial Hypertension
The goals of therapy in pulmonary arterial hypertension (PAH) include alleviation of symptoms, improved exercise capacity, improvement in quality of life (QOL), preservation of right ventricular (RV) function, and reduction of mortality risk.1,2 The European Society of Cardiology and the European Respiratory Society (ESC/ERS) 2015 guidelines propose several factors that aid in determining whether a person with PAH has a low, intermediate, or high risk of 1-year mortality. These factors include presence of right heart failure, progression of symptoms, syncope, World Health Organization functional class (WHO-FC), N-terminal pro b-type brain natriuretic peptide (NT-proBNP) levels, 6-minute walk distance (6MWD), cardiopulmonary exercise testing (CPET), imaging, and hemodynamic parameters (Table 11). According to the ESC/ERS guidelines, an important goal is achieving or maintaining a low-risk status. Low-risk status is characterized by good exercise capacity, QOL, RV function, and reduced risk of mortality (1-year mortality from PAH is less than 5% when patients are low risk).1 Therefore, the initial and ongoing assessment of patients with PAH includes determination of functional capacity, measurement of exercise capacity via 6MWD or CPET, and assessing RV function through measurement of NT-proBNP or by echocardiography. In addition, patients with PAH should undergo a comprehensive assessment for the presence of comorbidities and disease complications, presence of right heart failure, and the rate of disease progression. Evaluation of all prognostic information and therapy decisions should be made individually.1
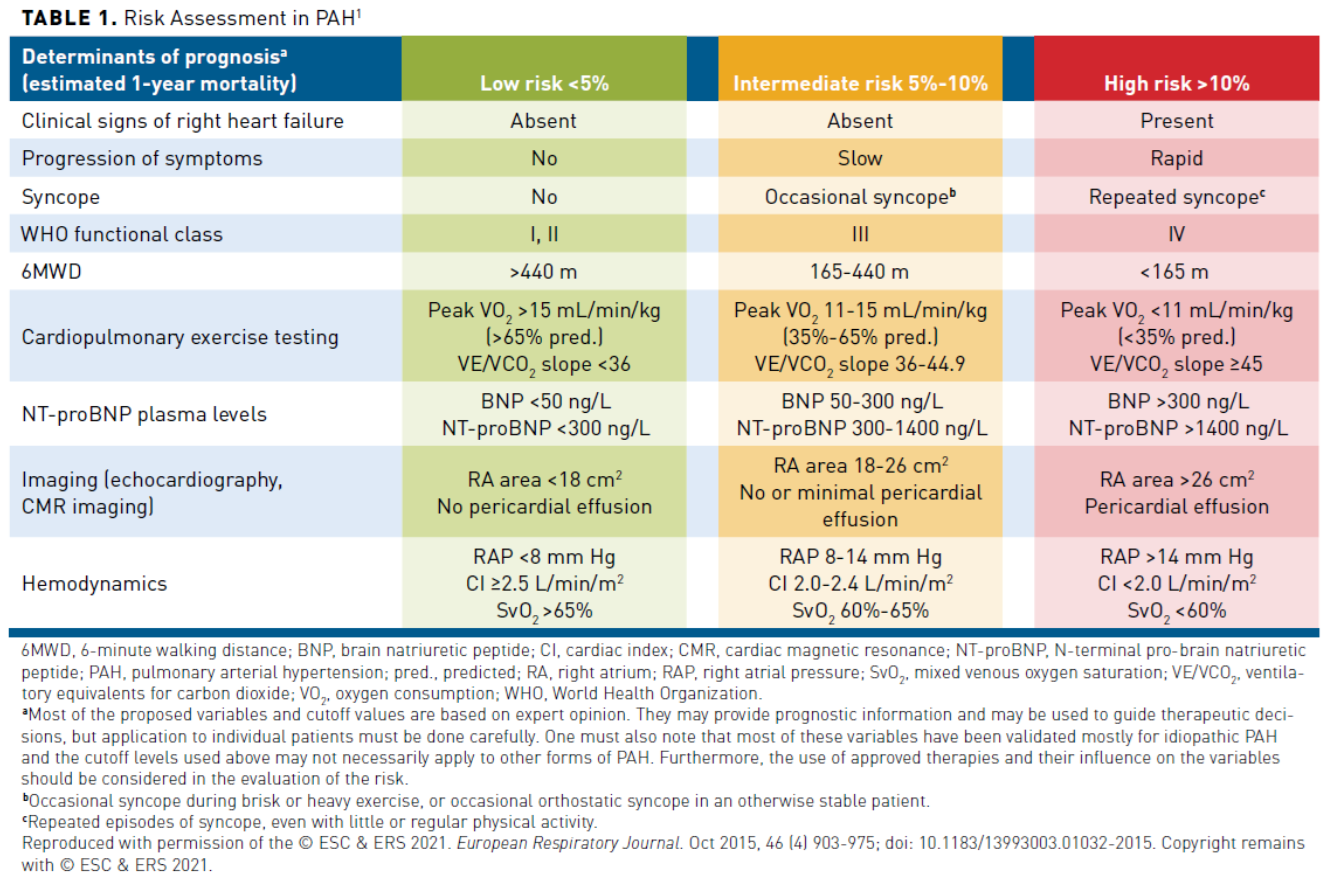
The 6-minute walking test is included in the PAH risk assessment and is the most widely adopted test. Several factors may affect 6MWD results including age, sex, height, weight, comorbidities, learning curve, and motivation. The threshold of greater than 440 meters is an acceptable goal for patients who are elderly and those with comorbidities, but a higher goal may be needed for younger, otherwise healthy patients. An improvement of 33 meters in 6MWD is considered clinically relevant by the American College of Chest Physicians (CHEST) guidelines. CPET provides important information about exercise capacity and cardiac function during exercise, such as peak oxygen consumption or uptake (peak VO2) that is mostly used in practice. CPET measurements add to the information provided by 6MWD. Variables measured in the CPET provide important prognostic information. Natriuretic peptides and brain natriuretic peptide (BNP)/NT-proBNP are markers of myocardial stress and have prognostic value in PAH. They are commonly used in clinical practice in diagnosis and follow-up assessments and as end points in clinical trials.1
Guidelines for the Pharmacologic Treatment of PAH
The approach to PAH treatment begins with nonpharmacologic management, which includes physical activity and rehabilitation, infection prevention, vaccinations against influenza and pneumococcal disease, counseling women with childbearing potential to avoid pregnancy as it may affect cardiopulmonary function, and counseling before travel. Supportive therapy is employed with oxygen when oxygen saturation is less than 90% or a PaO2 less than 60 mm Hg, loop diuretics, oral anticoagulation, and lifestyle modifications such as smoking cessation counseling and adherence to a low-sodium diet.1,2
The next step in initial management is vasoreactivity testing, which is recommended in patients with idiopathic PAH, heritable PAH, and drug- or toxin-induced PAH. This testing is done to determine whether high-dose calcium channel blockers (CCBs) are a treatment option for these individuals.1 Approximately 10% to 20% of patients will have a positive or vasoreactive response and are considered eligible for CCBs. Commonly used CCBs include nifedipine 30 mg daily or diltiazem 120 mg daily increased to the maximum tolerated dose over days to weeks, with monitoring of blood pressure, heart rate, and oxygen saturation during titration. Doses required for efficacy in PAH are often higher than those used for systemic hypertension (eg, amlodipine 20 mg daily, diltiazem 720 mg daily, nifedipine 240 mg daily). It is important to note that the efficacy of CCBs is short lived and 50% of patients will lose response within a year. Patients maintained on CCBs should therefore be reassessed after 3 to 6 months to determine if the CCB needs to be replaced with double or triple therapy.1
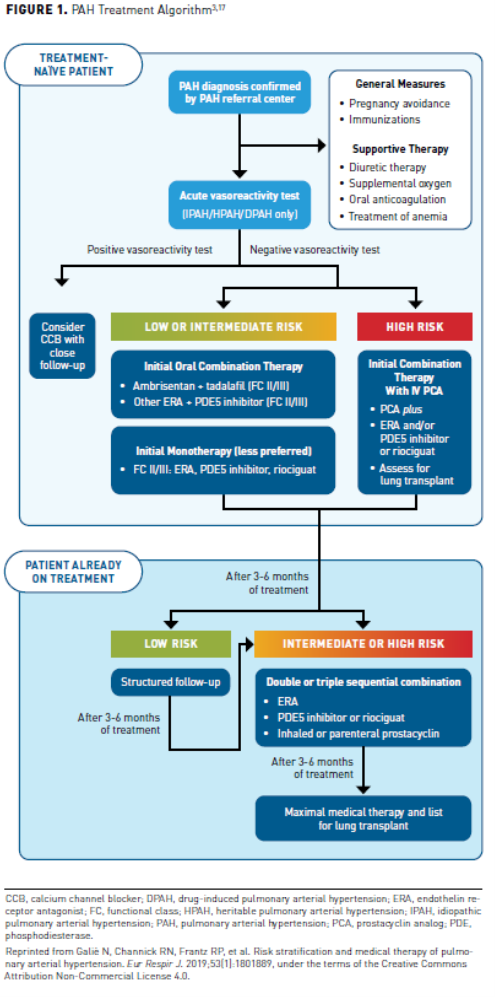
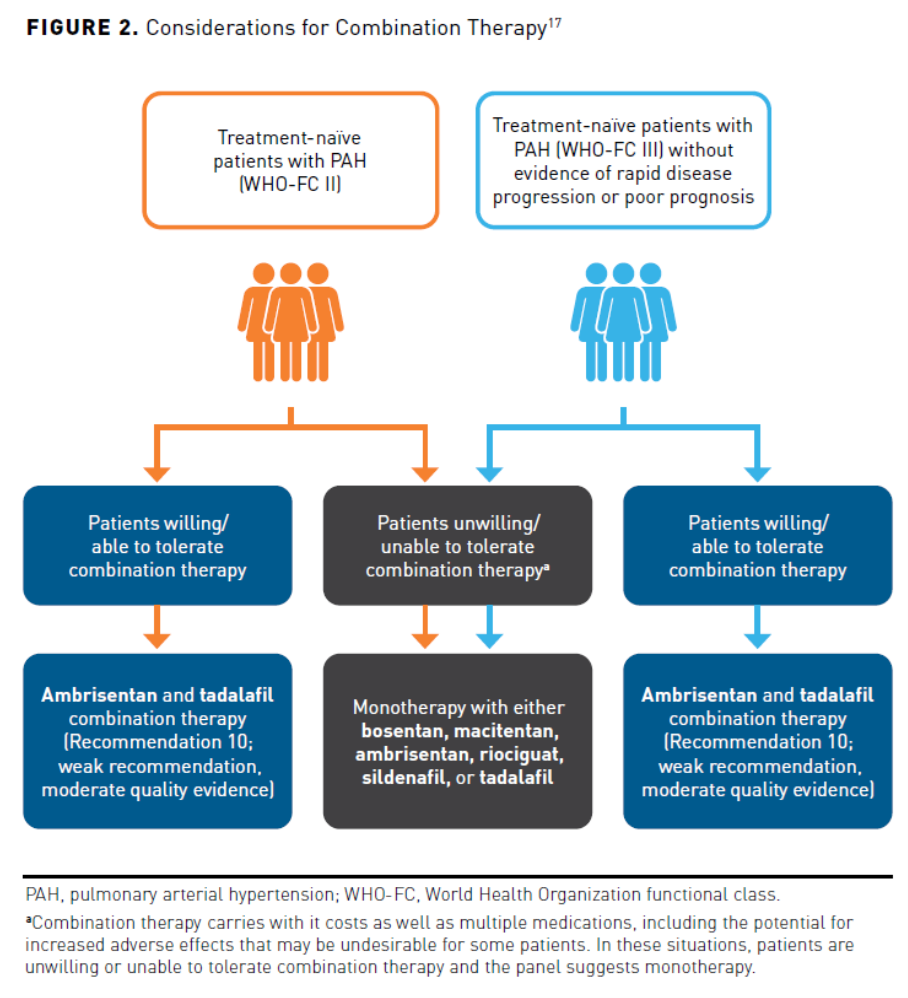
Individuals who are considered nonvasoreactive are not expected to benefit from CCBs and will be initiated on pharmacotherapy with single or dual combination therapy according to functional class and patient characteristics.1 Patients typically present in WHO-FC II and should be assessed for the risk of disease progression before therapy is initiated. Treatment is generally selected based on risk status, functional class, and patient preference. In treatment-naïve patients with low or intermediate risk, the preferred treatment is dual combination with an endothelin receptor antagonist (ERA) and a phosphodiesterase-5 (PDE5) inhibitor. The 2019 CHEST guidelines provide new recommendations for pharmacologic treatment in patients with WHO-FC II-III. A treatment algorithm that guides clinical decision making is provided based on patient functional class, disease progression, and response to treatment (Figure 13). The expert panel reviewed the literature and added recommendations to the treatment algorithm. The most important addition by the panel based on literature review was the recommendation to initiate combination treatment with ambrisentan and tadalafil in treatment-naïve patients with WHO-FC II-III to improve 6MWD (strong recommendation).
Pharmacologic Therapy for PAH
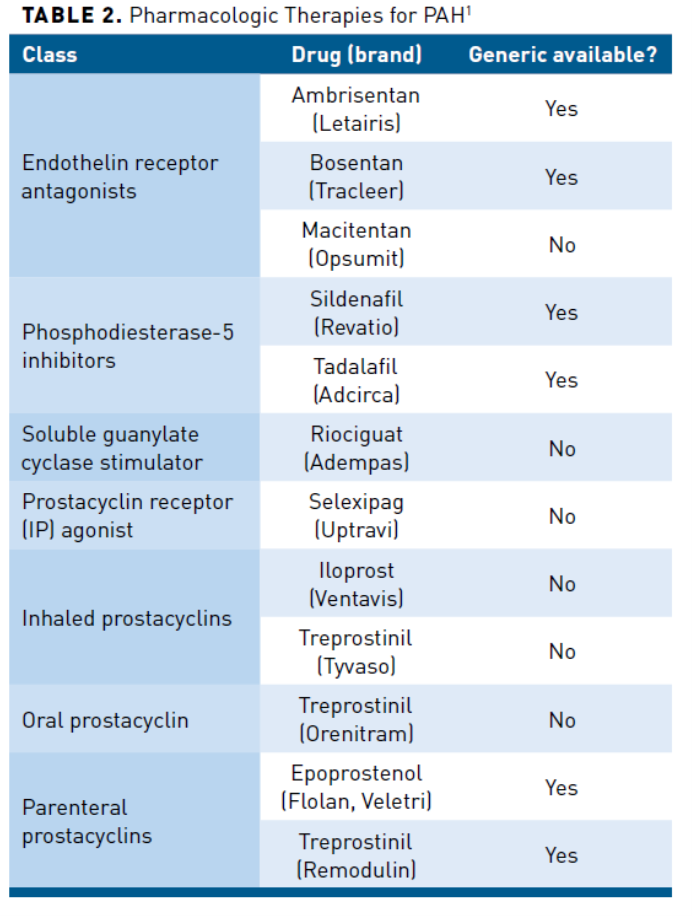
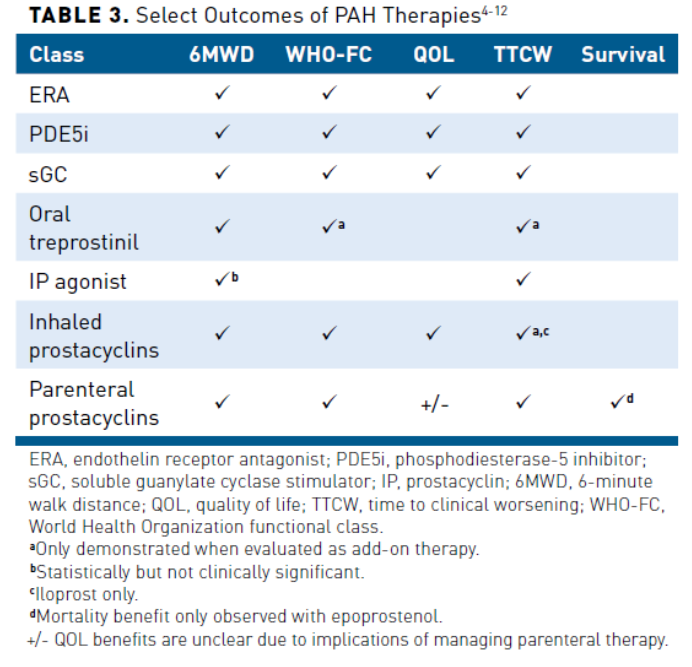
Pharmacologic therapy for PAH in those who are nonvasoreactive includes endothelin receptor agonists (ERAs) (ambrisentan, bosentan, and macitentan), PDE5 inhibitors (sildenafil and tadalafil), soluble guanylate cyclase stimulator (sGC) (riociguat), and prostacyclin therapies (epoprostenol, iloprost treprostinil, and selexipag) (Table 21).Phase 3 studies of PAH-specific therapies have evaluated and demonstrated varying outcomes (Table 34-12).
Combination therapy targets the multiple signaling pathways involved in PAH, resulting in improved hemodynamic parameters, symptomatic relief, and improved exercise capacity. Evidence from several clinical trials pointing to benefits of upfront combination therapy has shifted treatment of PAH, leading to changes in guidelines recommending this strategy in WHO-FC II-III patients who can tolerate it, with the strongest level of evidence for ambrisentan plus tadalafil.1 While ambrisentan and tadalafil is currently the only FDA-approved combination specifically endorsed by CHEST guidelines as initial therapy for patients with less severe symptoms, alternative combination treatments that have shown benefit in clinical trials include macitentan plus sildenafil (SERAPHIN), tadalafil plus bosentan (PHIRST), riociguat plus bosentan (PATENT-1 and -2), selexipag plus ERA or PDE5 inhibitor (GRIPHON), and oral treprostinil plus ERA, riociguat, or PDE5 inhibitor (FREEDOM-EV).5,6,8,13-16 Considerations for use of individual agents and evidence for double and triple therapy as well as emerging agents will be discussed below. Parenteral prostanoids (intravenous [IV] epoprostenol, IV or subcutaneous [SC] treprostinil) are recommended in patients with rapidly progressing disease or WHO-FC IV to improve 6MWD. In these patients, combination therapy should be started, which must include prostacyclin therapy.
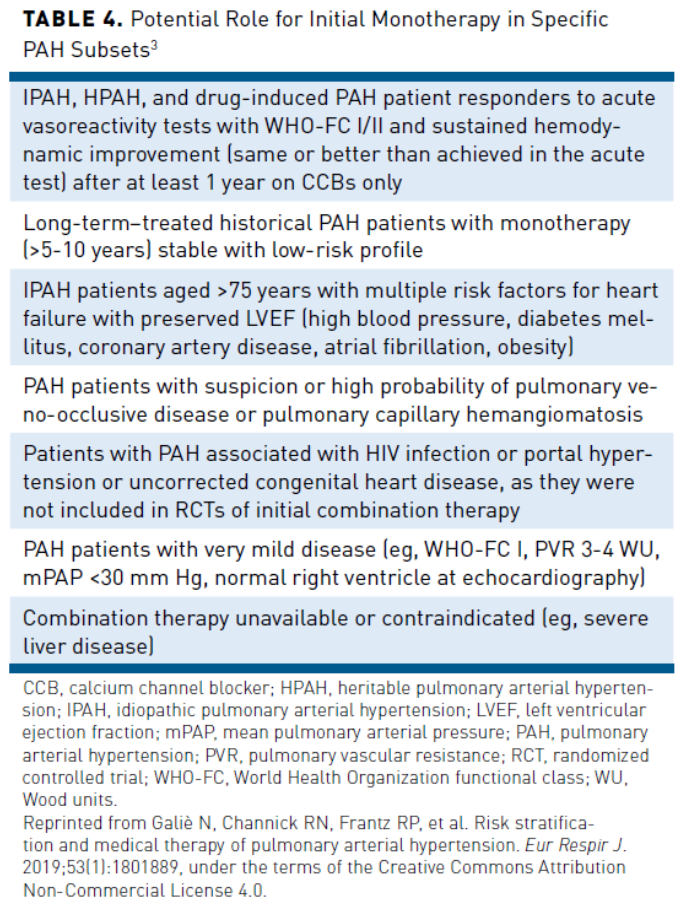
Patients who cannot tolerate combination treatment can receive monotherapy with ambrisentan, sildenafil (strong recommendation); bosentan, macitentan, tadalafil, or riociguat (consensus-based recommendation).17 In addition, single-agent monotherapy may be appropriate in patients with a low-risk profile and who have been stable for several years (>5-10 years), in patients with comorbidities with risk factors for heart failure or with contraindications to combination therapy such as severe liver disease; a complete list is included in Table 4.3 In patients with inadequate response to bosentan, ambrisentan, or inhaled prostanoids, addition of riociguat to therapy has been shown to improve 6MWD, delay time to clinical worsening, and improve FC.17 Additionally, sequential combination therapy can be considered if there is inadequate response with monotherapy.17
Endothelin Receptor Antagonists
Endothelin receptor antagonists block the action of endothelin, a powerful vasoconstrictor and mitogen for smooth muscle associated with greater PAH severity and poor prognosis. The ERAs bosentan, ambrisentan, and macitentan are all administered orally and indicated in patients with PAH with WHO-FC II to IV. Bosentan and macitentan are nonselective, dual-action ERAs, and ambrisentan is a selective antagonist of endothelin receptor A (how differences in selectivity affect clinical outcomes remains unclear). Bosentan is initiated at 62.5 mg twice daily for 4 weeks and then increased to 125 mg twice daily; ambrisentan is initiated at 5 mg daily and increased to 10 mg daily; and macitentan is administered at 10 mg daily. ERAs are effective in improving exercise capacity, WHO-FC, hemodynamic parameters, and time to clinical worsening. AEs for the ERAs include peripheral edema, anemia, nasal congestion, and hepatotoxicity requiring monitoring of liver function tests. Bosentan is the most hepatotoxic and requires monthly monitoring of liver function tests as part of its REMS program, while occasional monitoring is recommended with macitentan and ambrisentan. Peripheral edema is mostly seen with bosentan and ambrisentan and can be managed with diuretics. All 3 ERAs require REMS enrollment for use due to teratogenicity and are contraindicated in pregnancy. Female patients with childbearing potential should have monthly pregnancy tests and receive counseling regarding dual contraception.18
Nitric Oxide Pathway Modulators
PDE5 Inhibitors
The PDE5 inhibitors sildenafil and tadalafil and the guanylate cyclase stimulator riociguat all target the nitric oxide (NO)-cyclic guanosine monophosphate pathway (cGMP). PDE5 inhibitors prevent the breakdown of cGMP and increase the effects of NO leading to vasodilation and antiproliferative effects on vascular smooth muscle cells. Sildenafil improves exercise capacity, FC, and hemodynamic parameters (mean pulmonary arterial pressure [mPAP]) and is approved for the treatment of PAH at 20 mg three times daily, although doses up to 80 mg three times daily are often used in practice. Tadalafil 40 mg per day is FDA approved for PAH and was shown to increase 6MWD, time to clinical worsening, and health-related quality of life (HRQOL). Common adverse effects (AEs) include headaches and flushing. Patients may experience changes in vision, such as blue-tinted vision or sudden loss of vision, which warrant drug discontinuation. Additionally, there is a risk of systemic hypotension and both agents are contraindicated with nitrates and riociguat due to an increased risk of hypotension.18
Riociguat
Riociguat is a soluble guanylate cyclase (sGC) stimulator that enhances the production of NO through a direct stimulation of sGC and increase in intracellular cGMP. It is initiated at 1 mg three times daily and titrated by 0.5 mg every 2 weeks to a maximum dose of 2.5 mg three times daily. Riociguat improved exercise capacity, pulmonary vascular resistance (PVR), NT-proBNP, time to clinical worsening, and WHO-FC in patients with PAH.17,18 Switching from a PDE5 inhibitor to riociguat in patients who do not respond to PDE5 inhibitors was shown to improve exercise capacity and hemodynamics.19 Riociguat is generally well tolerated with common AEs including headache, dizziness, dyspepsia, peripheral edema, and hypotension. Riociguat is contraindicated in pregnancy due to risk of teratogenicity and requires Risk Evaluation and Mitigation Strategy (REMS) enrollment for use. Women of childbearing potential require pregnancy tests and counseling for use of dual contraceptive methods. Concomitant administration with PDE5 inhibitors is contraindicated due to the increased risk of hypotension and lack of evidence of positive benefit/risk ratio. Smoking status should be assessed while on therapy, as smoking reduces riociguat concentrations by 50% to 60% and may require higher doses. While not the focus of this review, riociguat is the only agent currently approved for WHO group 4 pulmonary hypertension, otherwise known as chronic thromboembolic pulmonary hypertension.17
Prostacyclin Analogs and Prostacyclin Receptor Agonists
Prostacyclin analogs (epoprostenol, treprostinil, iloprost) and the prostacyclin receptor agonist selexipag increase cyclic adenosine monophosphate and cause nonselective pulmonary vasodilation. These agents have antiplatelet, antithrombotic, anti-inflammatory, and antiproliferative effects on pulmonary endothelial tissue.18
Selexipag
Selexipag is the most recent prostacyclin agent to receive FDA approval. It is a nonprostanoid oral agent with high selectivity to the prostacyclin-receptor (IP) over prostanoid receptors and promotes vasodilation in pulmonary vasculature, inhibition of platelet aggregation, and causes antiproliferative effects on smooth muscle cells. Selexipag is approved for the treatment of PAH to delay disease progression and reduce the risk of hospitalization and has shown benefits in patients with WHO-FC II-III. Compared with other prostanoids, selexipag provides the convenience of oral, twice-daily administration, and may have fewer gastrointestinal AEs. The dose is initiated at 200 μg twice daily with incremental dose increases every week to the highest tolerable dose, up to a maximum of 1600 μg twice daily.20
In the randomized double-blind, placebo-controlled phase 3, event-driven, Prostacyclin (PGI2) Receptor Agonist in Pulmonary Arterial Hypertension (GRIPHON) trial, selexipag added to background therapy significantly reduced the risk of primary composite end point of death or complications related to PAH compared with placebo (HR, 0.6; 99% CI, 0.46-0.78; P <.001). The treatment effect was driven by differences in disease progression and hospitalization. Study patients had either no therapy or had a stable dose of ERA, PDE5 inhibitor, or both.16,17 The most common AEs included headache, diarrhea, and nausea that were mild to moderate in nature and consistent with AEs observed with other prostacyclin therapies. Other AEs included anemia and hyperthyroidism.16 GRIPHON was the largest randomized controlled study in patients with PAH with 1156 patients enrolled; the study showed no significant difference in mortality between study groups. Because the improvement in 6MWD did not meet a clinically significant threshold of 33 meters and a composite was used as a primary end point, these results were considered insufficient evidence by the CHEST guidelines to provide a recommendation for or against the use of selexipag, given the committee’s consideration of 6MWD as the clinically relevant outcome; of note, this is inconsistent with prior guidelines, which recommended selexipag as a monotherapy option in WHO-FC II-III and as add-on therapy for WHO-FC IV.16,17 Recent post hoc analysis of the GRIPHON trial presented at the American Thoracic Society (ATS) International Conference showed greater benefit associated with early initiation of selexipag. Patients who started treatment within 6 months of their PAH diagnosis had a 55% risk reduction in morbidity and mortality versus placebo, whereas those initiated after 6 months experienced a 30% risk reduction versus placebo. Additionally, a more pronounced treatment response was observed in patients treated earlier.13 Observational real-world studies from database registries of 250 patients taking selexipag 1 year or longer (SPHERE) showed that patients maintained on selexipag remained in the same risk group or improved to a less severe risk group. Patients treated earlier were more likely to be WHO-FC II and treatment naïve from Asia/Eastern Europe than those treated later.21
Iloprost
Iloprost is an inhaled formulation of prostacyclin for PAH indicated for WHO-FC III-IV. In studies, iloprost has been shown to improve exercise capacity and FC. Iloprost is administered via the Adaptive Aerosol Delivery inhalation system at a starting dose of 2.5 μg given 6 to 9 times per day (every 2 hours during waking hours) to achieve clinical effect due to its short half-life of 25 minutes. Its use is limited in practice due to the frequency of administration, which takes about 10 minutes per dose, and preference to initiate parenteral therapy in patients with more severe illness. It is well tolerated, and the main AEs include flushing and jaw pain. Additional AEs include cough and throat irritation due to its route of administration.1
Treprostinil
Treprostinil is FDA approved for patients with WHO-FC II-IV. It is available in parenteral (for IV or SC use), inhaled, and oral formulations.18
Oral Treprostinil in Combination With Background Therapies
Treprostinil diolamine oral formulation has been shown to improve exercise capacity as monotherapy in treatment-naïve patients with WHO-FC III.18,22 The treprostinil extended-release tablet is initiated at 0.125 mg three times daily with food or 0.25 mg twice daily with food, titrated in increments of 0.125 three times daily or 0.25-0.5 mg twice daily every 3 to 4 days as tolerated. The treprostinil dose should be adjusted in patients with hepatic impairment (Child-Pugh class A) and avoided in those with Child-Pugh class B liver impairment.22 Recent data from the pivotal trial FREEDOM-EV (A Phase III, International, Multi-Center, Randomized, Double-Blind, Placebo-Controlled, Clinical Worsening Study of UT-15C in Subjects With Pulmonary Arterial Hypertension Receiving Background Oral Monotherapy) showed that initiation of oral treprostinil reduced the risk of clinical progression by 25% in patients who were maintained on PAH monotherapy. Delayed time to clinical worsening in the oral treprostinil group was largely due to a 61% reduction in risk of disease progression (HR, 0.39; 95% CI, 0.23-0.66; P <.001). Administration of oral treprostinil decreased plasma levels of NT-proBNP and led to improvements in 6MWD and WHO-FC starting at week 12 through week 48. There were reduced requirements for initiation of inhaled or parenteral prostacyclin in the oral treprostinil group.8 The most common AEs were headache, diarrhea, flushing, nausea, vomiting, jaw pain, dizziness, pain in extremities, and myalgia.8 Additionally, a follow-up hemodynamics study was conducted in 61 patients who received right-heart catheterization at baseline and week 24. A 19% significant reduction in PVR (−134.09 vs −9.20; P = .0241) and 8% increase in cardiac output (0.42 vs −0.30; P = .0051) compared with placebo was demonstrated. There were no significant changes in mPAP, mean pulmonary arterial wedge pressure, or right atrial pressure.23 Importantly, the results of these studies have not yet been incorporated into current guideline recommendations.
Inhaled Treprostinil in Combination With Background Therapies
Inhaled treprostinil was shown to improve exercise capacity and QOL predominantly as add-on therapy in patients with WHO-FC III-IV. Inhaled treprostinil is recommended by the 2019 CHEST guidelines in combination with a PDE5 inhibitor and an ERA in patients with WHO-FC IV who cannot tolerate IV prostanoids.17 In the TRIUMPH-I (Double Blind Placebo Controlled Clinical Investigation Into the Efficacy and Tolerability of Inhaled Treprostinil Sodium in Patients With Severe Pulmonary Arterial Hypertension) trial, inhaled treprostinil was administered with a target dose of 54 µg four times daily as an add-on to background therapy bosentan or sildenafil. This benefit was sustained for 2 years after initiation.9,24,25 Treprostinil inhaled solution must be taken through the Tyvaso inhalation system in 4 daily sessions every 4 hours, at 3 breaths per treatment session (18 µg each). The dose is increased by 3 breaths per session at 1- to 2-week intervals to 9 breaths (54 µg each) per session as tolerated.24 Common AEs include throat irritation, cough, headache, nausea, dizziness, and flushing. Additionally, patients require monitoring for systemic hypotension, especially those on concomitant antihypertensives or vasodilators. Inhaled treprostinil recently demonstrated benefits related to 6MWD, NT-proBNP, and clinical worsening in WHO group 3 pulmonary hypertension secondary to interstitial lung disease and led to submission of a new drug application (NDA) for this indication.26
Parenteral Treprostinil
Treprostinil injection for SC or IV use is recommended in patients with WHO-FC III with rapid disease progression and patients in WHO-FC IV. Treprostinil has a half-life of about 4 hours, allowing more flexibility and lower risk of rebound pulmonary hypertension with discontinuation as compared with epoprostenol. It is prescribed for patients to diminish symptoms associated with exercise intolerance and in those who require transition from epoprostenol to reduce the rate of clinical deterioration. While parenteral treprostinil has not demonstrated survival benefits, this agent is often used interchangeably with epoprostenol based on patient-specific factors and institution preferences. The risks and benefits of each drug should be considered before selection and/or transition. Treprostinil is initiated at 1.25 ng/kg/min and titrated based on efficacy and tolerability. Patients being considered for parenteral therapy currently require both an ability to mix the prescribed drug and manage an external pump. The SC route is preferred to decrease risk of complications related to IV therapy, but the drug can be administered by central IV if the patient cannot tolerate the SC route. The CADD-Legacy 1 pump is an external device used for continuous IV delivery and provides up to 2 days’ supply of medication, while the more compact CADD-MS 3 pump used for SC administration provides a 72-hour supply of treprostinil. SC infusion may cause infusion-site pain and injection reactions that may warrant consideration of the IV route. Other AEs include headache, diarrhea, vasodilation, edema, jaw pain, and hypotension.27 As of March 2019, a generic treprostinil infusion had been approved for IV and SC use, although currently available SC delivery systems are not patented to deliver this formulation.28,29
Epoprostenol
Epoprostenol is a synthetic prostacyclin analog with a short half-life of 3 to 5 minutes requiring a continuous infusion. Epoprostenol is the first and only PAH-specific agent to have demonstrated survival benefits to-date.30 Additionally, IV epoprostenol was shown to improve exercise capacity and hemodynamics and is approved in patients with WHO-FC III-IV. Of note, there are currently 2 available formulations of epoprostenol available, Flolan and Veletri. Important differences in these formulations include the diluents used for reconstitution and resulting stability. Flolanoriginally required a glycine buffer diluent for a stability of 8 hours at room temperature, creating a need for ice packs while administering. Since then, a pH 12 sterile diluent has been developed, allowing for 72 hours of thermostability. Veletri may be mixed with sterile water or normal saline and is thermostable for up to 72 hours at room temperature, depending on concentration. Hospitalization is required for initiation of both epoprostenol and treprostinil. Epoprostenol must be administered via a pump through central venous catheter at a starting dose of 2 ng/kg/min and is increased to a target dose of 25 to 40 ng/kg/min based on efficacy and tolerability. The titration rate depends on disease severity, symptoms, and AEs; the maximal dose has not been established. AEs that may occur during titration and are common to all prostanoids include headache, nausea, vomiting and diarrhea, flushing, hypotension, and jaw pain.18,31 Severe AEs may occur with this complex delivery system and include infection, thrombosis, pump malfunction, and interruption of the infusion, which is considered a medical emergency. A backup supply of the drug and pump are required to ensure patient safety should the pump malfunction. The rate of central venous catheter infection was estimated at 2% per year and contributes to morbidity and mortality with continuous epoprostenol.18
Combination Therapies
Ambrisentan and Tadalafil
The Ambrisentan and Tadalafil in Patients with Pulmonary Arterial Hypertension (AMBITION) trial evaluated therapies with different intracellular target pathways (PDE5 inhibitor tadalafil 40 mg and ERA ambrisentan 10 mg) that did not have pharmacokinetic interaction. Results of AMBITION demonstrated that the upfront use of combination therapy with ambrisentan and tadalafil in patients with PAH who were treatment naïve significantly reduced the risk of clinical failure events compared with either ambrisentan or tadalafil monotherapy. The primary end point of the study was time to event of clinical failure defined as composite of death, hospitalization for worsening of PAH, disease progression, or unsatisfactory long-term clinical response until patients’ final assessment. Patients in the combination therapy group had a 50% lower risk of clinical failure than in the pooled monotherapy group (HR, 0.5; 95% CI, 0.35-0.72; P <.001), and had greater improvements in 6MWD and greater reductions from baseline in NT-proBNP than in the pooled monotherapy group (P <.001). Additionally, there was a 63% reduction in risk of PAH-related hospitalization when compared with pooled monotherapy (HR, 0.372; 95% CI, 0.217-0.639; P = .0002). The most common AEs in the combination group were peripheral edema, headache, nasal congestion, and anemia, but there was no difference in drug discontinuation due to AEs.32 Results of the AMBITION trial led to the combination being approved by the FDA as a first-line treatment in patients with WHO-FC II and prompted the updated CHEST guideline recommendations for the initial use of ambrisentan and tadalafil in PAH.17,32
Other Dual Combination Therapies
Combination treatment with alternative PDE5 inhibitors and ERAs has also been shown to be safe and effective, such as macitentan plus sildenafil and tadalafil plus bosentan; these were shown to improve clinical outcomes such as time to clinical failure in the SERAPHIN and PHIRST studies, respectively. Benefits have also been demonstrated with riociguat plus bosentan in the PATENT-2 study. In the GRIPHON trial, selexipag added to background therapy demonstrated a 49% decrease in clinical worsening regardless of type of background therapy (ERA, PDE5 inhibitor, or combination). While the ambrisentan and tadalafil combination is specifically endorsed by the guidelines, clinical experts consider these other combinations as appropriate treatment options. Currently, the CHEST guidelines make no specific recommendations for or against the use of selexipag.3,5,6,14,15
Not all evaluations of upfront combination therapy have demonstrated positive results. In the BREATH-2 study, the combination of IV epoprostenol plus bosentan was no better than epoprostenol alone.33 More recent evidence suggested this combination may improve hemodynamics but not clinical outcomes such as mortality.34 CHEST guidelines do not recommend routine addition of bosentan in patients when initiating parenteral prostacyclin therapy. While these guidelines make no specific recommendation for upfront combination therapy in patients with WHO-FC IV symptoms, they acknowledge the need for studies to evaluate preferred combination therapies in patients with advanced illness.17 The decision for which combination to use should include consideration of patient-specific factors (eg, risk assessment, preferences, tolerability). In general, upfront combinations of oral therapies discussed above are most appropriate in patients with low or intermediate risk (WHO-FC II-III without rapid progression), while consideration should be given for therapy that includes a parenteral prostacyclin in patients with high-risk features (Figure 217).
Triple Combination Therapy
Despite the known benefits of upfront dual therapy, the merits of triple combination therapy remain unclear. A study of 21 patients with newly diagnosed PAH treated with a combination of ambrisentan, tadalafil, and SC treprostinil was associated with improvements in hemodynamics, WHO-FC, and 6MWD at a median of 2 years follow-up.35 Similar results were also demonstrated in a pilot study evaluating upfront triple combination therapy with IV epoprostenol, bosentan, and sildenafil.36 While the results of this study were largely positive, it is difficult to draw conclusions based on its retrospective nature and lack of a comparator group.
More recently, the phase 3b TRITON study evaluated initial triple therapy with selexipag, macitentan, and tadalafil in comparison to initial dual therapy with macitentan and tadalafil in 247 patients with newly diagnosed PAH. Results of the study were recently presented at the ATS 2020 International Conference. Initial triple therapy reduced the risk of disease progression by 41% (HR, 0.59; 95% CI, 0.32-1.09, P = .087), and while there was an improvement in the primary end point of change in PVR and in secondary end points of 6MWD, NT-proBNP, and worsening of FC at week 26, these were nonsignificant. Reported AEs were consistent with known profile of drug classes (headaches, diarrhea, nausea, pain in extremities, jaw pain, and vomiting) and occurred in higher frequency in the triple therapy group. These results did not provide sufficient evidence to support upfront triple therapy and the benefit of adding selexipag remains unclear; however, the trend toward improved measures warrants further investigation. The full publication is awaited to assess whether initial triple therapy versus dual therapy improves long-term outcomes.37
Current guidelines make no specific mention of upfront triple combination therapy. Limitations of current evidence include lack of robust study designs and heterogenous nature of therapies evaluated (ie, oral vs parenteral). Special consideration should be given to patient preference in this setting, given the overwhelming nature of a new PAH diagnosis and the implications for adjusting to multiple new therapies, particularly as this relates to parenteral therapy. Given the limited data available, the role of this strategy remains unclear but may be considered for specific patients (ie, those with high-risk features willing and able to tolerate triple combination therapy).
Emerging Pharmacotherapeutic Modalities
Treprostinil Infusion Systems
The Remunity pump delivery system, initially cleared by the FDA in May 2019 for patients aged 22 years and older, provides continuous SC delivery for 3 days (72 hours). This system provides a compact design that is water resistant, simplicity of prefilled cassettes, and is programmable with a wireless remote and is approved for commercial distribution as of February 2021. The cassettes used in the pump are prefilled in a specialty pharmacy and delivered to the patient.27,38-40 An implantable system for Remodulin (ISR) is also under development that allows patients to receive IV prostacyclin therapy without an external pump or tubing, decreasing the risk of bloodstream infections, line malfunctions, and infusion-site pain. The pump is surgically implanted in the patient’s abdomen. The trial is ongoing with initial implants expected to occur this year in 2021.41 The Trevyent PatchPump system is a prefilled and preprogrammed disposable skin patch pump system that received the FDA Orphan Drug designation in 2016. It is designed to deliver treprostinil SC for 2 days (48 hours) with decreased site pain. The device manufacturer is awaiting review of the NDA submitted in 2019; a decision is expected later this year.42
Treprostinil dry powder inhalation
LIQ861 is a formulation of treprostinil administered through a disposable dry powder inhaler (DPI) using a novel technology that optimizes drug delivery into the lung. The delivery system provides 25 µg four times daily increased by 25 µg weekly until symptomatic relief as tolerated. The novel Particle Replication in Nonwetting Templates (PRINT) technology aims to improve current inhaled therapies and the DPI device has the potential to improve treatment satisfaction, offers convenience and improved QOL for patients with PAH, requiring 4 to 8 breaths per day compared with currently available treatment options that require 36 breaths per day and use of a nebulizer.43 LIQ861 was evaluated in the phase 3 open-label Investigation of the Safety and Pharmacology of Dry Powder Inhalation of Treprostinil (INSPIRE) trial in 121 patients transitioned from Tyvaso or stable on less than or equal to 2 nonprostacyclin therapies.43 At baseline, the majority of patients were female (81.8%) with the majority (66.1%) being in New York Heart Association (NYHA)-FC II and the rest in NYHA-FC III (33.9%). At 2 months follow up, 75.9% of patients maintained their NYHA-FC and 20.5% improved with similar results between patients transitioning from Tyvaso and in stable patient groups. Improvements were also noted in 6MWD and QOL scores. LIQ861 was evaluated at doses ranging from 26.5 µg to 159 µg with no serious AEs observed.44,45 Following an NDA submission, a complete response letter was issued from the FDA in November 2020.
Other dry powder formulations for treprostinil are currently under development, including treprostinil inhalation powder. Like LIQ861, the purpose of this formulation is to create convenience in administration and enhance the delivery of inhaled treprostinil. The DPI uses a Technosphere particle technology (TreT) allowing for a high concentration of the drug to be delivered into the lungs. Doses administered to healthy volunteers in a dosing study varied from 30 µg to 180 µg per dose and produced clinically relevant concentrations of treprostinil. TreT was well tolerated and the most frequently reported AEs were cough and headache.46 The safety and tolerability of TreT is currently being evaluated in the BREEZE study. This open-label, phase 1b study is evaluating the transition of patients on a stable dose of inhaled Tyvaso to an equivalent dose of DPI formulation for a minimum of 3 weeks. Outcomes of interest include pharmacokinetic data, 6MWD, safety, and patient satisfaction. BREEZE is anticipated to be completed in March 2022 with an NDA filing anticipated in April 2021.47
Ralinepag
Ralinepag is a selective nonprostanoid prostacyclin receptor agonist formulated as an oral, extended-release tablet taken once daily. In a phase 2 randomized placebo-controlled trial, ralinepag significantly reduced PVR compared with placebo (−29.8%; P = .03) in 61 patients receiving mono- (41%) or dual- (59%) background therapy. Ralinepag was initiated at a dose of 10 μg twice daily and titrated as tolerated over 9 weeks to a maximum total daily dosage of 600 μg (300 μg twice daily). Reported AEs included headache, nausea, diarrhea, jaw pain, and flushing.48,49 Ongoing phase 3 studies, part of the ADVANCE clinical program, are evaluating the effect of ralinepag on clinical outcomes and exercise capacity. ADVANCE OUTCOMES will assess the impact of once-daily ralinepag on morbidity and mortality, including disease progression, clinical worsening, and survival in 700 patients randomized 1:1 to receive ralinepag or placebo plus PAH-specific therapy.50 This trial is estimated to be completed in December 2021. The effect of ralinepag on exercise capacity after 28 weeks of treatment is being assessed in ADVANCE CAPACITY (using change in VO2 derived from CPET).49,51 The trial is currently recruiting and estimated to be completed in September 2023.
Sotatercept
Disruptions in the transforming growth factor (TGF)-β and bone morphogenetic protein receptor type II (BMPR2) pathways lead to cellular proliferation and vascular remodeling seen in PAH. Sotatercept is a first-in-class, selective ligand trap for members of the TGF-β superfamily (activins, growth differentiation factors, others) that can rebalance BMPR2 signaling and restore vascular homeostasis. It is administered as an SC injection every 3 weeks.52 Phase 2 results presented at the ATS 2020 conference showed sotatercept in combination with background therapies reduced mPAP, increased 6MWD, reduced NT-proBNP, and improved WHO-FC compared with placebo (N = 106). Notable AEs in the sotatercept group included a hemoglobin increase (n = 7) and thrombocytopenia (n = 7); other AEs included headache, diarrhea, peripheral edema, dizziness, fatigue, hypokalemia, and nausea.52-54 Sotatercept has received breakthrough therapy designation from the FDA and the ongoing STELLAR phase 3, randomized, double-blind, controlled trial is evaluating sotatercept 0.7 mg/kg SC every 3 weeks plus background therapy compared with placebo in 284 patients; it is planned for completion in December 2022. The primary end point of the trial is change from baseline in 6MWD with secondary outcomes including hemodynamic improvements, WHO-FC changes, time to death or clinical worsening, and changes in QOL scores.55
Conclusions
Recent developments in pharmacotherapy for the treatment of PAH led to new FDA approvals of agents such as ERAs, nitric oxide modulators, and prostacyclin therapies that target the underlying pathophysiology of the disease. Combination therapy with these agents has increased effectiveness and reduced the risk of death and disease progression. Survival is expected to increase with earlier initiation of combination therapies and application of an individualized treatment approach. Advances in pharmacotherapy and latest results from pivotal trials evaluating different agents in combination have shifted the treatment paradigm of PAH. Emerging therapies and novel delivery formulations provide new options to the therapeutic armamentarium of PAH management. These therapies promise added convenience, with the potential for reduced AEs and optimized clinical outcomes.
Author affiliation: Stormi Gale, PharmD, BCCP, BCPS, is assistant professor, Department of Pharmacy Practice and Science, University of Maryland School of Pharmacy, Baltimore, MD.
Funding source: This activity is supported by an educational grant from United Therapeutics Corporation.
Author disclosure: Dr Gale has no financial relationships with commercial interests to disclose.
Authorship information: Substantial contributions to analysis and interpretation of data, drafting of the manuscript, and critical revision of the manuscript for important intellectual content.
Address correspondence to: sgale@rx.umaryland.edu
Medical writing and editorial support: Marielle Fares, PharmD, BCGP
REFERENCES
1. Galiè N, Humbert M, Vachiery JL, et al. 2015 ESC/ERS guidelines for the diagnosis and treatment of pulmonary hypertension: the Joint Task Force for the Diagnosis and Treatment of Pulmonary Hypertension of the European Society of Cardiology (ESC) and the European Respiratory Society (ERS): endorsed by: Association for European Paediatric and Congenital Cardiology (AEPC), International Society for Heart and Lung Transplantation (ISHLT). Eur Respir J. 2015;46(4):903-975. doi: 10.1183/13993003.01032-2015
2. Moote R, Attrdige RL, Levine DJ. Pulmonary arterial hypertension. Pharmacotherapy: A Pathophysiologic Approach, 10e. DiPiro JT, Talbert RL, Yee GC, Matzke GR, Wells BG, Posey M, eds. 2017; McGraw-Hill Education.
3. Galiè N, Channick RN, Frantz RP, et al. Risk stratification and medical therapy of pulmonary arterial hypertension. Eur Respir J. 2019;53(1):1801889. doi: 10.1183/13993003.01889-2018
4. Simonneau G, Rubin LJ, Galiè N, et al; PACES Study Group. Addition of sildenafil to long-term intravenous epoprostenol therapy in patients with pulmonary arterial hypertension: a randomized trial. Ann Intern Med. 2008;149(8):521-530. doi: 10.7326/0003-4819-149-8-200810210-00004
5. Pulido T, Adzerikho I, Channick RN, et al; SERAPHIN Investigators. Macitentan and morbidity and mortality in pulmonary arterial hypertension. N Engl J Med. 2013;369(9):809-818. doi: 10.1056/NEJMoa1213917
6. Ghofrani HA, Galiè N, Grimminger F, et al; PATENT-1 Study Group. Riociguat for the treatment of pulmonary arterial hypertension. N Engl J Med. 2013;369(4):330-340. doi: 10.1056/NEJMoa1209655
7. Tapson VF, Torres F, Kermeen F, et al. Oral treprostinil for the treatment of pulmonary arterial hypertension in patients on background endothelin receptor antagonist and/or phosphodiesterase type 5 inhibitor therapy (the FREEDOM-C study): a randomized controlled trial. Chest. 2012;142(6):1383-1390. doi: 10.1378/chest.11-2212
8. White RJ, Jerjes-Sanchez C, Bohns Meyer GM, et al; FREEDOM-EV Investigators. Combination therapy with oral treprostinil for pulmonary arterial hypertension: a double-blind placebo-controlled clinical trial. Am J Respir Crit Care Med. 2020;201(6):707-717. doi: 10.1164/rccm.201908-1640OC
9. McLaughlin VV, Benza RL, Rubin LJ, et al. Addition of inhaled treprostinil to oral therapy for pulmonary arterial hypertension: a randomized controlled clinical trial. J Am Coll Cardiol. 2010;55(18):1915-1922. doi: 10.1016/j.jacc.2010.01.027
10. Olschewski H, Simonneau G, Galiè N, et al; Aerosolized Iloprost Randomized Study Group. Inhaled iloprost for severe pulmonary hypertension. N Engl J Med. 2002;347(5):322-329. doi: 10.1056/NEJMoa020204
11. Barst RJ, Rubin LJ, Wong WA, et al; Primary Pulmonary Hypertension Study Group. A comparison of continuous intravenous epoprostenol (prostacyclin) with conventional therapy for primary pulmonary hypertension. N Engl J Med. 1996;334(5):296-301. doi: 10.1056/NEJM199602013340504
12. Simonneau G, Barst RJ, Galiè N, et al; Treprostinil Study Group. Continuous subcutaneous infusion of treprostinil, a prostacyclin analogue, in patients with pulmonary arterial hypertension: a double-blind, randomized, placebo-controlled trial. Am J Respir Crit Care Med. 2002;165(6):800-804. doi: 10.1164/ajrccm.165.6.2106079
13. Gaine SP, Sitbon O, Channick RN, et al. The impact of time from diagnosis at baseline on long-term outcome in the GRIPHON study: selexipag in pulmonary arterial hypertension. Am J Resp Crit Care Med. 2020;201:A2502. Accessed February 21, 2021. atsjournals.org/doi/abs/10.1164/ajrccm-conference.2019.199.1_MeetingAbstracts.A2502
14. Rubin LJ, Galiè N, Grimminger F, et al. Riociguat for the treatment of pulmonary arterial hypertension: a long-term extension study (PATENT-2). Eur Respir J. 2015;45(5):1303-1313. doi: 10.1183/09031936.00090614
15. Galiè N, Brundage BH, Ghofrani HA, Oudiz RJ, Simonneau G, et al; Pulmonary Arterial Hypertension and Response to Tadalafil (PHIRST) Study Group. Tadalafil therapy for pulmonary arterial hypertension. Circulation. 2009;119(22):28942893. doi: 10.1161/CIRCULATIONAHA.108.839274
16. Sitbon O, Channick R, Chin KM, et al; GRIPHON Investigators. Selexipag for the treatment of pulmonary arterial hypertension. N Engl J Med. 2015;373(26):2522-2533. doi: 10.1056/NEJMoa1503184
17. Klinger JR, Elliott CG, Levine DJ, et al. Therapy for pulmonary arterial hypertension in adults:
update of the CHEST Guideline and Expert Panel Report. Chest. 2019;155(3):565-586.
doi: 10.1016/j.chest.2018.11.030
18. Thenappan T, Ormiston ML, Ryan JJ, Archer SL. Pulmonary arterial hypertension: pathogenesis and clinical management. BMJ. 2018;360:j5492. doi: 10.1136/bmj.j5492
19. Hoeper MM, Simonneau G, Corris PA, et al. RESPITE: switching to riociguat in pulmonary arterial hypertension patients with inadequate response to phosphodiesterase-5 inhibitors. Eur Respir J. 2017;50(3):1602425. doi: 10.1183/13993003.02425-2016
20. Uptravi. Prescribing information. Actelion Pharmaceuticals US, Inc; 2015. Accessed January 1, 2021. www.accessdata.fda.gov/drugsatfda_docs/label/2015/207947s000lbl.pdf
21. Farber HW, Chakinala MM, Highland KB, et al. Risk assessment at baseline and one year in patients with pulmonary arterial hypertension (PAH): data from the first 250 patients enrolled in SPHERE (Uptravi [selexipag]: the Users Drug registry). Am J Respir Crit Care Med. 2019;199:A2503.
22. Orenitram. Prescribing information. United Therapeutics Corp; 2019. Accessed February 19, 2021. orenitramhcp.com/pdf/Orenitram_Full_Prescribing_Information.pdf
23. White RJ, Meyer G, Pulido T, et al. Treatment with oral treprostinil improves hemodynamics in participants with PAH. Poster presented at Pulmonaty Vascular Research Institute Annual World Congress 2021. Accessed February 19, 2021. pvrinstitute.org/media/8383/0097-jim-white.pdf
24. Tyvaso. Prescribing information. United Therapeutics Corp; 2017. Accessed February 21, 2021. www.tyvaso.com/hcp/pdf/TYVASO-PI.pdf?v=102017
25. Benza RL, Seeger W, McLaughlin VV, et al. Long-term effects of inhaled treprostinil in patients with pulmonary arterial hypertension: the Treprostinil Sodium Inhalation Used in the Managmenet of Pulmonary Arterial Hypertension (TRIUMPH) study open-label extension. J Heart Lung Transplant. 2011;30(12):1327-1333. doi: 10.1016/j.healun.2011.08019
26. Waxman A, Restrepo-Jaramillo R, Thenappan T, et al. Inhaled treprostinil in pulmonary hypertension due to interstitial lung disease. N Engl J Med. 2021;384(4):325-334. doi: 10.1056/NEJMoa2008470
27. Remodulin. Prescribing information. United Therapeutics Corp; 2018. Accessed January 1, 2021. www.remodulin.com/files/remodulin-pi.pdf
28. Sandoz exclusively launches treprostinil injection, the first generic version of Remodulin, in the US. News release. Sandoz. March 26, 2019. Accessed January 27, 2020. www.us.sandoz.com/news/media-releases/sandoz-exclusively-launches-treprostinil-injection-first-generic-version
29. Pulmonary Hypertension Association. Generic treprostinil injection launched for intravenous use. April 4, 2019. Accessed January 21, 2021.https://phassociation.org/generic-treprostinil-injection-launched-for-intravenous-use/#:~:text=Generic%20treprostinil%2C%20the%20pulmonary%20arterial,both%20intravenous%20and%20subcutaneous%20use
30. Barst RJ, Rubin LJ, Long WA, et al; Primary Pulmonary Hypertension Study Group. A comparison of continuous intravenous epoprostenol (prostacyclin) with conventional therapy for primary pulmonary hypertension. N Engl J Med. 1996;334(5):296-301. doi: 10.1056/NEJM199602013340504
31. Veletri. Prescribing information. Actelion Pharmaceuticals US, Inc; 2012. Accessed January 1, 2021. www.accessdata.fda.gov/drugsatfda_docs/label/2012/022260s005lbl.pdf
32. Galiè N, Barbera JA, Frost AE, et al; AMBITION Investigators. Initial use of ambrisentan plus tadalafil in pulmonary arterial hypertension. N Engl J Med. 2015;373(9):834-844. doi: 10.1056/NEJMoa1413687
33. Humbert M, Barst RJ, Robbins IM, et al. Combination of bosentan with epoprostenol in pulmonary arterial hypertension: BREATHE-2. Eur Respir J. 2004;24(3):353-359. doi: 10.1183/09031936.04.00028404
34. Kemp K, Savale L, O’Callaghan DS, et al. Usefulness of first-line combination therapy with epoprostenol and bosentan in pulmonary arterial hypertension: an observational study. J Heart Lung Transplant. 2012;31(2):150-158. doi: 10.1016/j.healun.2011.11.00
35. D’Alto M, Badagliacca R, Argiento P, et al. Risk reduction and right heart reverse remodeling by upfront triple combination therapy in pulmonary arterial hypertension. Chest. 2020;157(2):376-383. doi: 10.1016/j.chest.2019.09.009
36. Sitbon O, Jais X, Savale V, et al. Upfront triple combination therapy in pulmonary arterial hypertension: a pilot study. Eur Respir J. 2014;43(6):1691-1697. doi: 10.1183/09031936.00116313
37. Chin KM, Sitbon O, Doelberg M, et al. Efficacy and safety of initial triple oral versus initial double oral combination therapy in patients either newly diagnosed pulmonary arterial hypertension (PAH): results of the randomized controlled TRITON study. Am J Respir Crit Car Med. 2020;201:A2928. Accessed February 21, 2021. abstractsonline.com/pp8/#!/8998/presentation/17706
38. Remodulin.com. Pump options: CADD-Legacy 1 for patients starting IV pump therapy with Remodulin. Accessed January 1, 2021. remodulin.com/pump-options/cadd-legacy/
39. Remunity pump for Remodulin (treprostinil) injection. October 19, 20219. Accessed February 21, 2021. remodulin.com/files/remunity-isi.pdf
40. Biospace. United Therapeutics and DEKA announce additional FDA clearance related to the Unity subcutaneous delivery system for Remodulin. February 24, 2020. Accessed February 21, 2021. biospace.com/article/releases/united-therapeutics-and-deka-announce-additional-fda-clearance-related-to-the-unity-subcutaneous-delivery-system-for-remodulin-/
41. Implantable System for Remodulin Post-Approval Study (ISR PAS). ClinicalTrials.gov. Updated April 24, 2020. Accessed February 15, 2021. clinicaltrials.gov/ct2/show/NCT03833323
42. Pulmonary Hypertension Association. New PAH treatment system submitted for FDA approval. October 3, 2019. Accessed December 31, 2020. phassociation.org/new-pah-treatment-system-submitted-for-fda-approval/#:~:text=The%20FDA%20received%20a%20New,to%20be%20marketed%20to%20consumers
43. Liquidia releases final LIQ861 results from pivotal phase 3 INSPIRE study in patients with pulmonary arterial hypertension. News release. Intrado. April 30, 2020. Accessed December 31, 2020. globenewswire.com/news-release/2020/04/30/2025110/0/en/Liquidia-Releases-Final-LIQ861-Results-from-Pivotal-Phase-3-INSPIRE-Study-in-Patients-with-Pulmonary-Arterial-Hypertension.html
44. Hill NS, Feldman JP, Sahay S, et al; INSPIRE Investigators. INSPIRE: A phase 3 open-label, multicenter study to evaluate the safety and tolerability of LIQ861 in pulmonary arterial hypertension (PAH) (Investigation of the Safety and Pharmacology of Dry Powder Inhalation of Treprostinil NCT03399604). Exploratory Efficacy Endpoints Analysis at Month 2. Am J Respir Crit Care Med. 2020;201:A
45. Investigation of the Safety and Pharmacology of Dry Powder Inhalation of Treprostinil (INSPIRE). ClinicalTrials.gov. Updated November 6, 2020. Accessed December 31, 2020. clinicaltrials.gov/ct2/show/NCT03399604
46. Smith P, Watkins C, Kraft K, Grant M. A phase 1, single-center, open-label, dose-rising clinical trial to evaluate the pharmacokinetics, safety and tolerability of treprostinil inhalation powder (TreT) in healthy normal volunteers. Eur Respir J. 2019;54:PA4749. doi: 10.1183/13993003.congress-2019.PA4749
47. Open-label, Clinical Study to Evaluate the Safety and Tolerability of TreT in Subjects With PAH Currently Using Tyvaso (BREEZE). ClinicalTrials.gov. Updated December 24, 2020. Accessed February 15, 2021. clinicaltrials.gov/ct2/show/NCT03950739
48. Torres F, Farber H, Ristic A, et al. Efficacy and safety of ralinepag, a novel oral IP agonist, in PAH patients on mono or dual background therapy: results from a phase 2 randomised, parallel group, placebo-controlled trial. Eur Respir J. 2019;54(4):1901030. doi: 10.1183/13993003.01030-2019
49. McLaughlin VV, Channick R, Walsh B, et al. Study design of the phase 3 ADVANCE Program evaluating time-to clinical events and exercise capacity in patients with pulmonary arterial hypertension treated with ralinepag. Am J Respir Crit Care Med. 2019;199:A5074.
50. A Study Evaluating the Efficacy and Safety of Ralinepag to Improve Treatment Outcomes in PAH Patients. ClinicalTrials.gov. Updated December 21, 2020. Accessed January 2, 2021. clinicaltrials.gov/ct2/show/NCT03626688
51. A Study of Ralinepag to Evaluate Effects on Exercise Capacity by CPET in Subjects With WHO Group 1 PH (CAPACITY). ClinicalTrials.gov. Updated January 22, 2021. Accessed February 21, 2021. clinicaltrials.gov/ct2/show/NCT04084678
52. Badesch D, Gibbs SJ, Gomberg-Maitland M, et al. PULSAR: a phase 2, randomised, double-blind, placebo-controlled study to assess the efficacy and safety of sotatercept (ACE-011) when added to standard of care (SOC) for treatment of pulmonary arterial hypertension (PAH). European Respiratory Society 2019 International Congress. Abstract 19918. Eur Respir J. 2019;54:PA4750.
53. Biospace. Acceleron presents topline results of the PULSAR phase 2 trial of sotatercept in patients with pulmonary arterial hypertension. June 24, 2020. Accessed December 30, 2020. biospace.com/article/releases/acceleron-presents-topline-results-of-the-pulsar-phase-2-trial-of-sotatercept-in-patients-with-pulmonary-arterial-hypertension/
54. A Study of Sotatercept for the Treatment of Pulmonary Arterial Hypertension (PAH) (PULSAR). ClinicalTrials.gov. Updated July 12, 2020. Accessed December 30, 2020. clinicaltrials.gov/ct2/show/NCT03496207
55. A Study of Sotatercept for the Treatment of Pulmonary Arterial Hypertension (STELLAR). ClinicalTrials.gov. Updated February 1, 2021. Accessed February 21, 2021. clinicaltrials.gov/ct2/show/NCT04576988
