- Center on Health Equity & Access
- Clinical
- Health Care Cost
- Health Care Delivery
- Insurance
- Policy
- Technology
- Value-Based Care
Ixekizumab Improves Children’s Dermatology Life Quality Index Over 2 Years
Patients with moderate-to-severe plaque psoriasis experienced early and sustained improvements through 108 weeks of treatment.
Pediatric patients with moderate-to-severe plaque psoriasis showed early and sustained improvements in each domain of the Children’s Dermatology Life Quality Index (CDLQI) and Dermatology Life Quality Index (DLQI) through 108 weeks of treatment with ixekixumab, according to a poster presented at the Society of Pediatric Dermatology (SPD) annual conference in Toronto, Canada.1
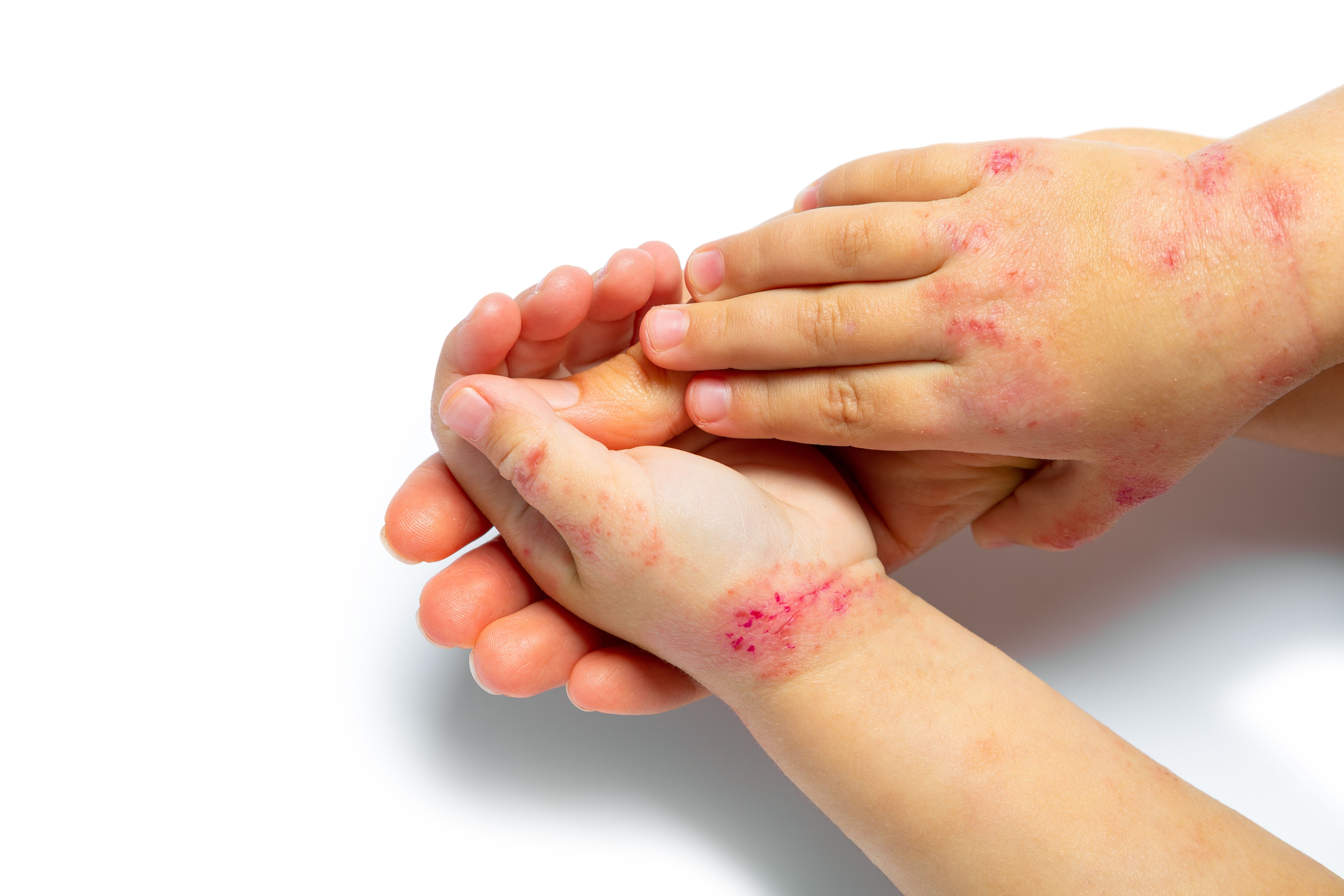
Plaque psoriasis can greatly impact the health-related quality of life in pediatric patients. Ixekizumab is a high-affinity monoclonal antibody that targets IL-17A, and is used to treat inflammatory disease, including plaque psoriasis. In a previous head-to-head study, ixekizumab outperformed adalimumab in terms of improving joint and skin disease in patients with psoriatic arthritis.2
In the current study, researchers aimed to evaluate the responses in each domain of the CDLQI and DLQI in children and adolescents treated with ixekizumab through 108 weeks.1
The study included a total of 171 patients who were randomized in a double-blind induction period to receive either ixekizumab (n = 115) or placebo (n =56). All patients participated in an open-label maintenance and extension period through 108 weeks.
Patients were between age 6 to 17 years and were candidates for phototherapy or systemic treatment or considered inadequately controlled by topical therapies. Additionally, concomitant therapies were allowed, including topical steroids, shampoo, moisturizers/emollients, bath oil, and oatmeal bath preparations. Furthermore, patients had to have a diagnosis of moderate-to-severe PsO for 6 or more months.
Exclusion criteria included prior use of etanercept or an IL-17 pathway inhibitor, and prior use of other therapies within specified time frames.
The researchers utilized the CDLQI for patients aged 6 to 16 years, and the DLQIO for patients aged 17 years. Each questionnaire had a total of 10 questions pertaining to 6 domains, with each questions scored 0 = not at all; 1 = only a little; 2 = quite a lot; and 3 = very much. Additionally, the questionnaires were evaluated on an overall scale of 0-30 with higher numbers associated with a larger effect on the patient’s life over the past week.
The CDLQI domain included symptoms and feelings, leisure, school or holidays, personal relationships, sleep, and treatment. The DLQI domain included symptoms and feelings, leisure, work and school, personal relationships, daily activities, and treatment.
The analysis population included all patients randomized to ixekizumab at week 0, who continued to receive ixekizumab through week 108 (n = 94). Additionally, the researchers observed data shown at each timepoint from weeks 0 through 108, including mean (SD) change from baseline for each domain of the CDLQI and DLQI, and the proportion of patients achieving CDLQI/DLQI 0/1.
Of the included participants, the mean (SD) age was 14.1 (2.9) years, with 54 (57.4%) females. Patients treated with ixezkizumab experienced improvements in each domain of the CDLQI (n = 67) and DLQI (n = 25) as early as week 4, with further reductions observed through week 108. Additionally, the proportion of patients who reported that plaque psoriasis had no effect on their life, according to CDLQI/DLQI 0/1, increased from 6.5% at baseline to over 60% of patients by 6 week 12, which increased to 76% by week 108.
Therefore, the researchers believe the study supports ixekizumab to provide early and sustained improvements in the quality of life for children and adolescents with moderate-to-severe plaque psoriasis.
References
1. Pinter A, Bobonich M, Swanson E, et al. Children’s dermatology life quality index responses in pediatric patients with moderate-to-severe plaque psoriasis treated with ixekizumab over a 2-year period – Results from the randomized IXORA-PEDS trial. Poster presented at the Society of Pediatric Dermatology (SPD 2024); July 11-24, 2024; Toronto Canada
2. Davio K. Head-to-head study shows ixekizumab outperforms adalimumab in psoriatic arthritis. The Center for Biosimilars®. October 3, 2019. https://www.centerforbiosimilars.com/view/headtohead-study-shows-ixekizumab-outperforms-adalimumab-in-psoriatic-arthritis
The Breakdown: Breast Cancer Research Awareness Day
August 19th 2025Breast cancer is the second most common cancer among women and the second leading cause of cancer-related deaths among women in the US. In light of Breast Cancer Research Awareness Day, The American Journal of Managed Care® breaks down the most recent advancements in breast cancer prevention, screening, and therapies.
Listen
The Importance of Examining and Preventing Atrial Fibrillation
August 29th 2023At this year’s American Society for Preventive Cardiology Congress on CVD Prevention, Emelia J. Benjamin, MD, ScM, delivered the Honorary Fellow Award Lecture, “The Imperative to Focus on the Prevention of Atrial Fibrillation,” as the recipient of this year’s Honorary Fellow of the American Society for Preventive Cardiology award.
Listen
